|
A BCC cell contains 2 atoms or other kind of particle.įor cubic crystals AP. 1 Characteristics of various types of structures are shown in the table below. Up to 24 cash back atomic packing factor APF or packing fraction is the fraction of volume in a crystal structure that is occupied by atoms. 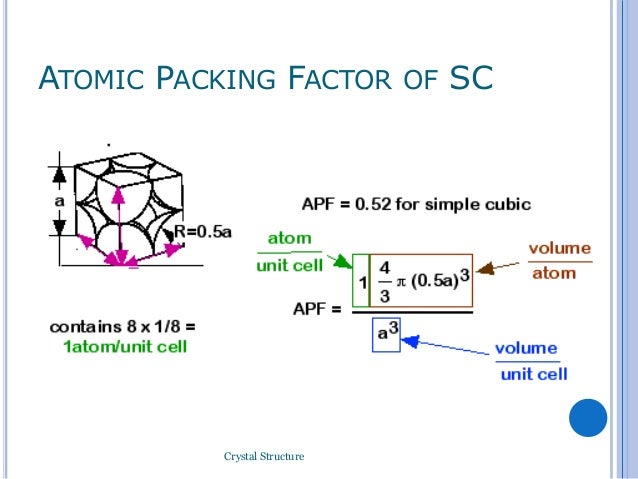 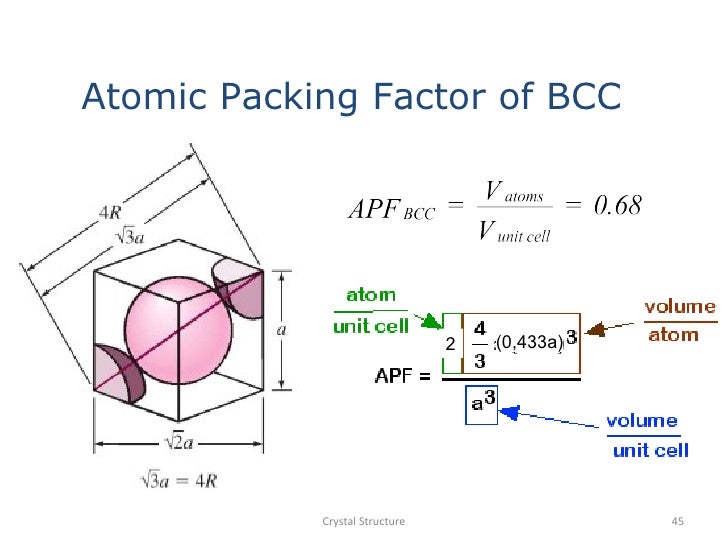
Atomic packing factorHCP lattice In the Hexagonal unit cell number of atoms 12 corner atoms x 16 shared by six unit cells Two face atoms x 12 3 interior 6. Calculate the atomic packing factor of BCC crystal structure. The atomic packing factor is defined as the ratio of the volume occupied by the average number of atoms in a unit cell to the volume of the unit cell. It can be defined as the ratio between the volume of the basic atoms of the unit cell which represent the volume of all atoms in one unit cell to the volume of the unit cell it self. It is dimensionless and always less than unity. With the help of the XRD pattern of the NaCl material given in the figure the unit cell parameters can be determinedcalculate.į its depends on the riadus of atoms and characrtiziation of chemical bondings. Mathematically Atomic Packing Factor APF. 
It is a dimensionless quantity and always less than unity. Pin On My Show that the atomic packing factor for BCC is 068 and forHCP is 074. FCC structure have APF Atomic Packing Factor of.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
